How Cells Clear Damaged Proteins: New Insights from Yeast

Cells rely on a system called ubiquitination to remove damaged or unwanted proteins—a process essential for health and survival. A key player in this system is a group of enzymes called ubiquitin ligases.
In a new study, Eric Fischer and his team focused on Tom1, a ubiquitin ligase found in yeast that also serves as a model for similar enzymes in humans. Tom1 has many jobs, including helping cells respond to DNA damage and tagging faulty proteins or free histones for removal.
Using advanced imaging (cryo-electron microscopy), the Fischer Lab captured Tom1 in the act of tagging proteins. They discovered that parts of the enzyme outside its core catalytic region are crucial for its function. In particular, they found a previously unknown site where Tom1 binds to a helper ubiquitin—this interaction helps ensure the correct assembly of a special chain that marks proteins for degradation.
Importantly, these newly identified regions of Tom1 are conserved in humans, and mutations in them are linked to disease, highlighting their biological importance.
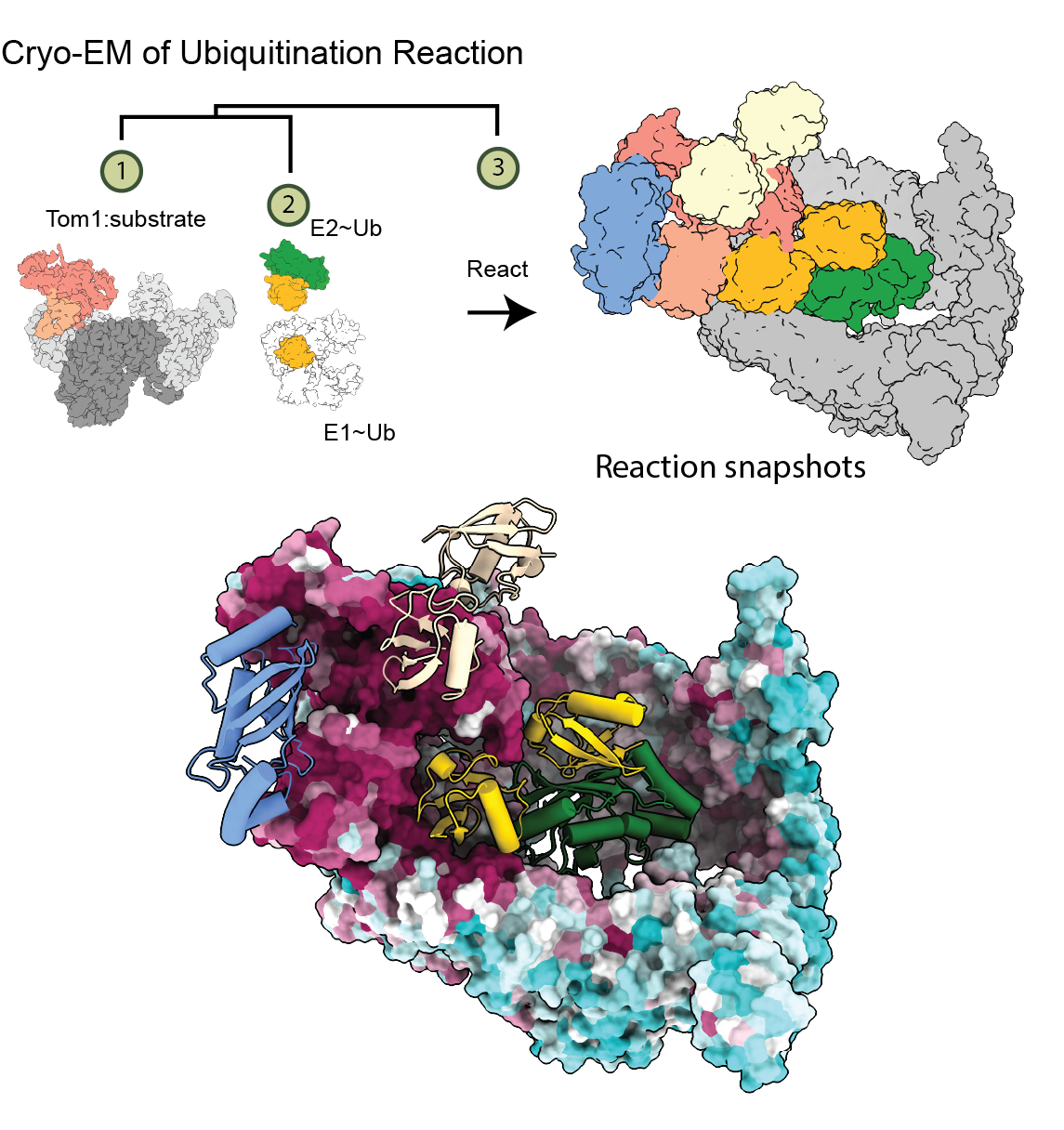
Schematic of the process to image Tom1 in the act of tagging proteins and the resulting overall structure highlighting conserved regions in dark purple and the helper ubiquitin in yellow.
Read the full article in the May 27, 2025 issue of Cell Reports.
Cover photo provided by Sam Ogden, courtesy of Dana-Farber Cancer Institute


